 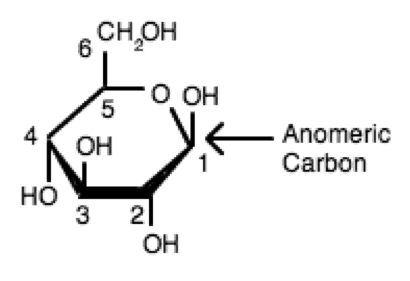
This explains why we can get energy from the starch in potatoes and other plants but not from cellulose, even though both starch and cellulose are polysaccharides composed of glucose molecules linked together. If the hydroxyl group on the anomeric carbon and the hydroxyl group on the chiral center furtherest away from the anomeric carbon are on the OPPOSITE side of the ring structure. At the anomeric carbon (the carbon that was an aldehyde or ketone in. When an alcohol adds to a carbonyl carbon. Anomers are pairs of stereoisomers that are formed when a simple linear sugar cyclizes. Molecules that are not mirror images but differ in spatial arrangements of atoms are diastereomers. They can be understood in terms of handedness, like gloves for the right or left hands. Enantiomers differ at the configuration of every stereocenter. The difference between the α and the β forms of sugars may seem trivial, but such structural differences are often crucial in biochemical reactions. stereoisomers that differ in configuration at only one C center. Enantiomers are stereoisomers that are non-superimposable mirror images. Any group written to the right in a Fischer projection appears below the plane of the ring in a Haworth projection, and any group written to the left in a Fischer projection appears above the plane in a Haworth projection. The structure is simplified to show only the functional groups attached to the carbon atoms. Glycoside formation locks the anomeric carbon such that. Condensation can occur with alcohols, amines, and amides. Glycosides: A general term referring to monosaccharide derivatives in which the functional group involving the anomeric carbon has an acetal or ketal structure. The molecules are drawn as planar hexagons with a darkened edge representing the side facing toward the viewer. Biological systems can tell the difference between stereoisomers. Nine of the nineteen l-amino acids commonly found in proteins are dextrorotatory (at a wavelength of 589 nm), and d-fructose is also referred to as levulose because it is levorotatory.\)) the cyclic forms of sugars are depicted using a convention first suggested by Walter N. Rather, it says that the compound's stereochemistry is related to that of the dextrorotatory or levorotatory enantiomer of glyceraldehyde-the dextrorotatory isomer of glyceraldehyde is, in fact, the d- isomer. The d/l labeling is unrelated to (+)/(−) it does not indicate which enantiomer is dextrorotatory and which is levorotatory. On the other hand, glycine, the amino acid derived from glyceraldehyde, has no optical activity, as it is not chiral (achiral). One example is the chiral amino acid alanine, which has two optical isomers, and they are labeled according to which isomer of glyceraldehyde they come from. Because.We know that a intramolecular ring is more 'comfortable' when the bond angles are larger (this is due to steric strain of the molecules). If the OH group is on the right hand side, the monosaccharide is of form. There is a chance that other OH's will attack, but they would be less stable and the major product will be a 6-sided ring. the configuration of the chiral carbon the carbonyl group determines whether a monosaccharide is D or L. In this system, compounds are named by analogy to glyceraldehyde, which, in general, produces unambiguous designations, but is easiest to see in the small biomolecules similar to glyceraldehyde. In aldoses (e.g., glucose) the two anomers differ in configuration at C1 (carbon having CHO group), while in ketoses (e. a hormone that stimulates the liver to release glucose into the bloodstream when levels are low. Anomers are the two stereoisomers that result from different alternative stereochemistry at its anomeric center. Certain chemical manipulations can be performed on glyceraldehyde without affecting its configuration, and its historical use for this purpose (possibly combined with its convenience as one of the smallest commonly used chiral molecules) has resulted in its use for nomenclature. The stereoisomers -D-glucopyranose and -D-mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position.The hydroxy group in -D-glucopyranose is equatorial (in the 'plane' of the ring), while in -D-mannopyranose the C-2 hydroxy group is axial (up from the 'plane' of the ring). Glyceraldehyde is chiral, and its two isomers are labeled d and l (typically typeset in small caps in published work). The d/l system (named after Latin dexter and laevus, right and left) names molecules by relating them to the molecule glyceraldehyde. During cyclization, the carbon in the carbonyl group converts into a new stereocenter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
